Description
Currently, women in rural areas in developing countries must travel to urban health centers or hospitals to receive certain birth control options that require insertion by skilled healthcare providers. The time and cost of travel presents a barrier to many women in rural location seeking long-term birth control options.
Problem
Ethiopia has a lack of trained healthcare providers—2.5 physicians per 100,000 people—limiting the delivery of effective long-term contraception including subcutaneous contraceptive implants.
Solution
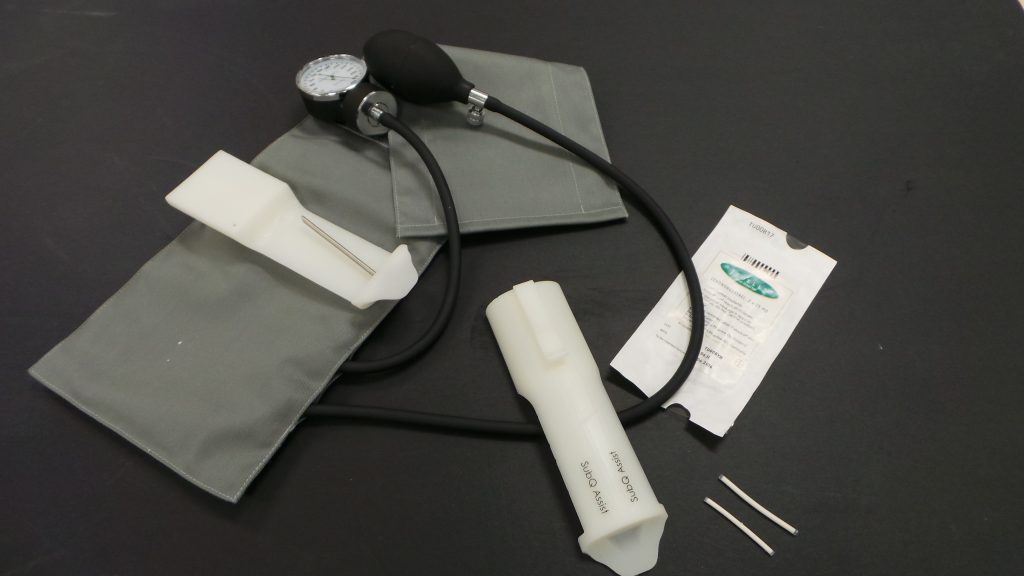
We co-designed, with Ethiopian healthcare providers and researchers, an assistive task-shifting device for accurate contraceptive implant administration.
Innovation
The assistive device, SubQ Assist, acts like a template, guiding healthcare providers during implant insertion, eliminating the possibility of unsafe administration and effectively shifting the task of administration to community health workers (CHWs).
Based in Evidence
In benchtop and cadaver testing, the accuracy of SubQ Assist (2.4 x 0.6 mm) was greater than the current contraceptive implant administration method (2.9 0.5 mm) with the target depth being 1.7 0.5 mm. In pre-clinical usability testing, 131 Ethiopian healthcare providers, including 53 CHWs, were recorded using the assistive device and were interviewed about their experience with the assistive device. Through the recordings and interviews, we identified that interest exists in SubQ Assist because it shifts the task of contraceptive implant administration and received feedback to inform design changes.
Expected Impact
In Ethiopia, over 30,000 CHWs service over 50% of the population, nearly 50 million people. In enabling CHWs to administer implants, we will provide contraceptive implants to 1.5 million women in Ethiopia. This increase in contraceptive implant use will prevent unintended pregnancies, abortions, miscarriages, and maternal and infant deaths. CHWs will track contraceptive implant use in patient medical records, which are already in use in Ethiopia.
Future Work
Currently, we are looking for partnering organizations to help develop and execute a small scale clinical trial in Ethiopia to demonstrate safety and efficacy. Additionally, we are seeking funding to support this trial and manufacturing and distribution partners.
Project Gallery
-
 University of Michigan biomedical engineering graduate student Ibrahim Mohedas present an advanced prototype of a sevise to aid insertion of long-term contraceptive to medical doctors at St. Paul Hospital in Addis Ababa, Ethiopia. Mohedas took feedback on the device that he hopes can help women in rural Ethiopia better control their birth giving. Once inverted under the skin, the contraceptive prevents pregnancy for three years. Thursday, January 29th, 2015.
Photo by Marcin Szczepanski/Senior Multimedia Producer, University of Michigan, College of Engineering
University of Michigan biomedical engineering graduate student Ibrahim Mohedas present an advanced prototype of a sevise to aid insertion of long-term contraceptive to medical doctors at St. Paul Hospital in Addis Ababa, Ethiopia. Mohedas took feedback on the device that he hopes can help women in rural Ethiopia better control their birth giving. Once inverted under the skin, the contraceptive prevents pregnancy for three years. Thursday, January 29th, 2015.
Photo by Marcin Szczepanski/Senior Multimedia Producer, University of Michigan, College of Engineering
-
 University of Michigan biomedical engineering graduate student Ibrahim Mohedas present an advanced prototype of a sevise to aid insertion of long-term contraceptive to medical doctors at St. Paul Hospital in Addis Ababa, Ethiopia. Thursday, January 29th, 2015.
Photo by Marcin Szczepanski/Senior Multimedia Producer, University of Michigan, College of Engineering
University of Michigan biomedical engineering graduate student Ibrahim Mohedas present an advanced prototype of a sevise to aid insertion of long-term contraceptive to medical doctors at St. Paul Hospital in Addis Ababa, Ethiopia. Thursday, January 29th, 2015.
Photo by Marcin Szczepanski/Senior Multimedia Producer, University of Michigan, College of Engineering
-

I have seen the people that my device will affect, and have met with the practitioners that will ultimately use it. The tangibility of that encounter makes me want to be a more accountable and ethical engineer.
What to Know About The Global Health Design Initiative

Program History
GHDI has been working with stakeholders for more than eight years to identify and address global health design challenges. Learn more about our history and core values.
Learn More →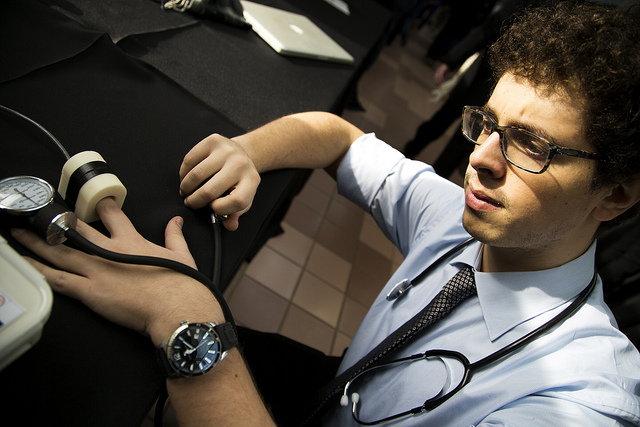
Project Outcomes
Since inception, we have worked on projects in maternal health, family planning, minimally invasive surgery, physical medicine and rehabilitation, and more. Explore our past and current projects.
Learn More →
Get Involved
There are many ways to get involved in global health work with GHDI. From taking on a need statement, to participating in an opportunity, or becoming a partner.
Learn More →